Data Snapshot
UW Medicine Hospitals:
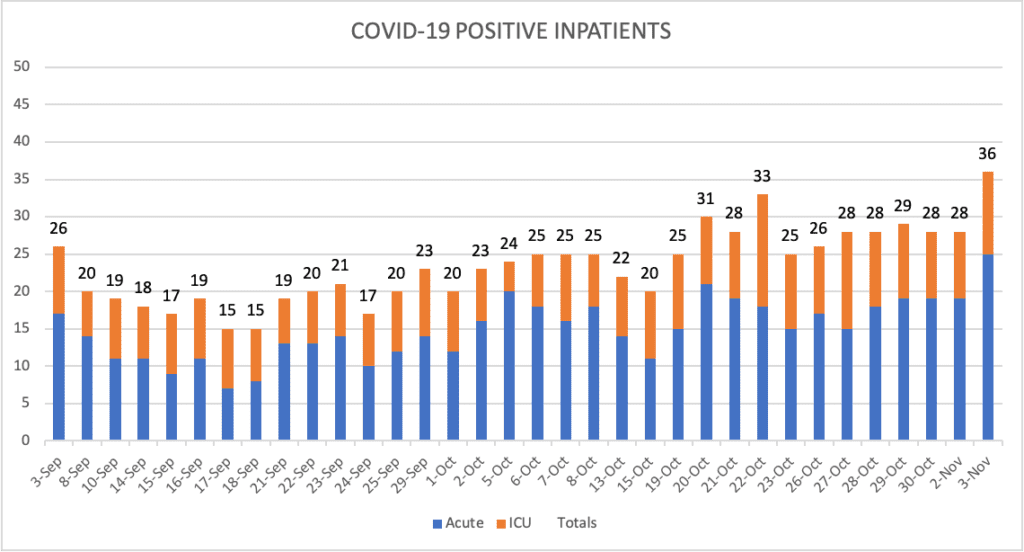
King County: The county reported 295 new positive cases and 2 new deaths on Nov. 2.
Washington: The state reported 109,354 cases and 2,378 deaths as of Nov. 1.
United States: The CDC reports 9,182,628 cases and 230,383 deaths as of Nov. 2.
Global: WHO reports 46,840,783 confirmed COVID-19 cases and 1,204,028 deaths as of Nov. 3.
*Numbers update frequently, please follow links for most up-to-date numbers.
COVID-19 Literature Situation Report
COVID-19 Literature Situation Report is a daily (M-F) newsletter put together by the Alliance for Pandemic Preparedness that provides a succinct summary of the latest scientific literature related to the COVID-19 pandemic.
Key Takeaways: COVID-19 Literature Situation Report Oct. 30, 2020
- Oxygen saturation did not decline among adults over age 65 years while they wore non-medical face masks. More.
- SARS-CoV-2 infections were found in 53% of household contacts of index cases in Tennessee and Wisconsin, with similar risk of secondary infection between index cases who were adults and those under age 18 years. 75% of secondary infections occurred within 5 days of the index patient’s onset of illness and only 40% of secondary cases were symptomatic at the time of detection by RT-PCR. More.
- Healthcare personnel in Minnesota who were working in congregate living or long-term care settings were less likely to wear appropriate PPE, more likely to work while symptomatic, and more likely to receive a positive test result during 14-day post-exposure monitoring, compared with healthcare personnel working in acute care settings. More.
UW Medicine in the News
Forbes: Travel Alert: Coronavirus Spreading Like Wildfire, Per Harvard-Brown Covid-19 Tracker
Featuring: IHME
“We are currently in the early stages of the third and largest surge of the pandemic. The influential model from the Institute for Health Metrics and Evaluation (IHME) at the University of Washington School of Medicine projects this surge will be longer and deadlier than the first two, not reaching its peak until mid-January. By February 1, 2021, the illness is expected have claimed nearly 400,000 American lives. Looking ahead at the next few weeks, public health experts are increasingly concerned about those planning to visit friends and family over Thanksgiving weekend, normally one of the busiest travel periods of the year. The recently issued detailed guidance for Thanksgiving from the Centers for Disease Control and Prevention deems “attending large indoor gatherings with people from outside of your household” to be high risk. In addition, a growing number of state governors are now urging residents to nix their Thanksgiving travel plans.”
The New York Times: Will the Hardest-Hit Communities Get the Coronavirus Vaccine?
Featuring: Beth Bell, Global Health
“It is an idea that may never have been tried in wide-scale vaccine distribution: Citing principles of equity and justice, experts are urging that people living in communities hardest-hit by the pandemic, which are often made up of Black and Hispanic populations, get a portion of the first, limited supply of coronavirus vaccines set aside just for them. A committee of experts advising Dr. Robert R. Redfield, the director of the Centers for Disease Control and Prevention, is considering the idea. But as it comes into focus, its underlying concepts and execution must be further defined, and the approach may then face legal and political challenges, even as the medical system grapples with the anticipated logistical hurdles of distributing new vaccines. The group that is formulating the allocation plan is the Advisory Committee on Immunization Practices, which consists of experts in medicine or public health who advise the C.D.C. on schedules for providing vaccine schedules for diseases like flu or chickenpox.
It will discuss a variety of ethical issues during its next meeting on Friday, but members say it will not vote on a final proposal until a vaccine receives either full approval or an emergency use authorization from the Food and Drug Administration, likely weeks or months from now. There are currently four vaccines in late-stage trials in the U.S.”
The Hospitalist: Two COVID-19 outpatient antibody drugs show encouraging results
Featuring: Deborah Fuller, Microbiology
“Deborah Fuller, PhD, professor in the Department of Microbiology at the University of Washington School of Medicine in Seattle, told Medscape Medical News the findings are «exciting» but only part of the treatment solution. ‘What’s remarkable about these two studies and others I’ve seen,’ she said, ‘is how consistent they are in terms of the window of time they will be effective, and that’s because they are just targeting the virus itself. They do not have an effect on the inflammation unless they stop the replication early enough.’ The treatments are effective when they are given near the time of diagnosis, she pointed out. ‘Once the virus has started that inflammatory cascade in your body, then that train has left the station and you have to deal with the inflammation,’ Fuller said. She says future treatments will likely have to include both the antiviral and anti-inflammatory properties, and physicians will have to assess what’s best, given the stage of the the patient’s disease.”
Tweet of the Week
If you missed Dr. Geoffrey Baird on @60Minutes last night, you can watch the segment here https://t.co/BrEPCr6igI@UWVirology @UWMedicine #COVID19 https://t.co/ynkw8zS84d
— UW Dept. of Laboratory Medicine and Pathology 🔬 (@uwlabmedpath) November 2, 2020

